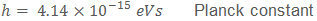The Planck relation
In 1900, German physicist Mac Planck was attempting to understand how the energy of radiation emitted by heated objects depends on frequency. He found that he was able to accurately describe this relationship if he assumed that the radiation was only released in small bundles of energy (an idea later known as quantisation), and that the energy of these bundles was directly proportional to their frequency.
The energy E of the bundles could therefore be written as...
The energy E of the bundles could therefore be written as...
...where f is the frequency of the radiation and h is a constant called the Planck constant.
Written in SI units the value of the Planck constant is:
Due to its extremely tiny value, the Planck constant is sometimes written using the units of eVs ("electron volt seconds"). An electron volt is defined as the amount of energy required to move an electron through an electric potential difference of 1 volt. The Planck constant written in eVs is:
Max Planck initially viewed his quantisation of energy as "a purely formal assumption ... actually I did not think much about it...". It took another German physicist, Albert Einstein, to realise that this quantisation was in fact a fundamental property of light.