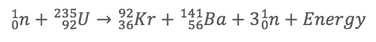Nuclear reactions
A nuclear reaction involves the collision of two nuclei (or a nucleus and a subatomic particle) to form one or more new nuclei.
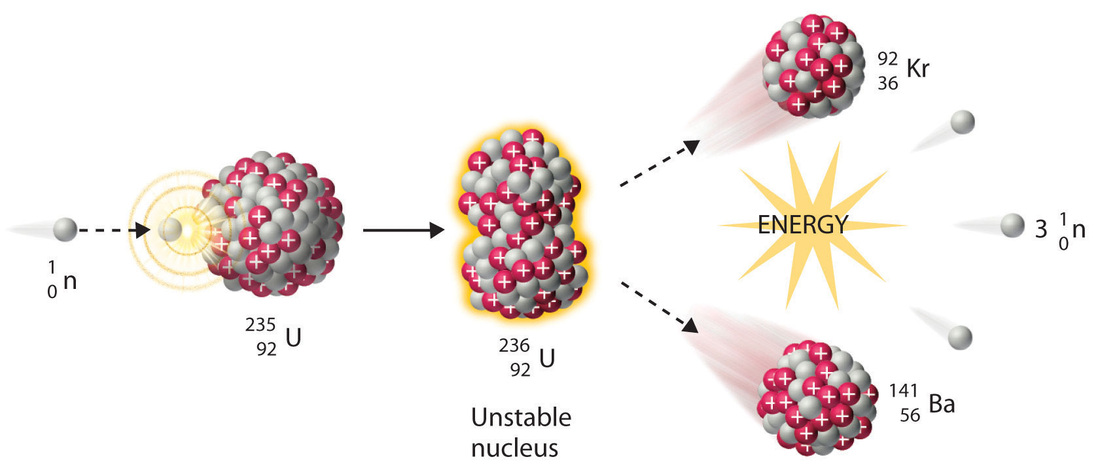
In the example below...
In the example below...
- A Uranium-235 nucleus is bombarded with a neutron.
- The two merge together to form an unstable isotope of Uranium, Uranium-236.
- Because Uranium-236 is unstable, it breaks apart into Krypton-92, Barium-141, and 3 neutrons.
Nuclear reactions can be described by equations much like those used to describe chemical reactions. The reaction above can be represented as follows...
 Click for larger image
Click for larger image
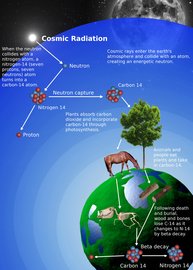
Nuclear reactions occur naturally all throughout nature. A barrage of high energy particles constantly rain down onto earth's surface from outer space (over 10,000 per square metre per second). Their collisions with nuclei in our atmosphere initiate nuclear reactions, producing unstable isotopes such as carbon-14, the mechanism of radiocarbon dating. Nuclear reactions also provide the fuel for all the stars in our universe, including our own sun.
Nuclear reactions can also be artificially created. Unstable isotopes produced from nuclear reactions in particle accelerators are used to diagnose and treat many diseases such as cancer. Nuclear power plants use the immense amount of heat produced by sustained nuclear reactions to generate electricity.
Nuclear reactions can also be artificially created. Unstable isotopes produced from nuclear reactions in particle accelerators are used to diagnose and treat many diseases such as cancer. Nuclear power plants use the immense amount of heat produced by sustained nuclear reactions to generate electricity.
Conservation laws in nuclear reactions
In a nuclear reaction there are several properties that are conserved. They include:
- Mass number
- Atomic number (charge)
- Momentum
- Mass (taking into account the mass of both matter and energy)