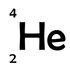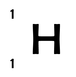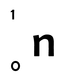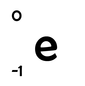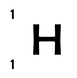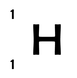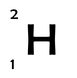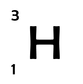Introduction to atoms and nuclei
Atoms are the basic building blocks of matter. A combination of atoms is called a molecule. The air you currently breathe is composed of many different types of molecules such as carbon dioxide. Carbon dioxide is made up of two oxygen atoms joined with one carbon atom.
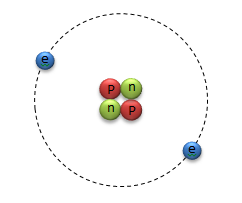
Atoms are made of 3 kinds of particles; positively charged protons, negatively charged electrons, and neutrally charged (uncharged) neutrons.
The central region of the atom is a tightly packed bundle of protons and neutrons called the nucleus (the plural of nucleus is nuclei). Because protons and neutrons reside in the nucleus, they are referred to as nucleons.
Electrons orbit around the nucleus at discrete distances that depend on how much energy they have.
We can symbolise an atom in the following way:
Atoms are made of 3 kinds of particles; positively charged protons, negatively charged electrons, and neutrally charged (uncharged) neutrons.
The central region of the atom is a tightly packed bundle of protons and neutrons called the nucleus (the plural of nucleus is nuclei). Because protons and neutrons reside in the nucleus, they are referred to as nucleons.
Electrons orbit around the nucleus at discrete distances that depend on how much energy they have.
We can symbolise an atom in the following way:
For example, chlorine (symbol Cl) has 17 protons and 18 neutrons; it therefore has a mass number of 35 and an atomic number of 17. We write it as:
Helium has 2 protons and 2 neutrons; it therefore has a mass number of 4 and an atomic number of 2. We write it as:
Hydrogen has only 1 proton and 0 neutrons; it therefore has both a mass number of 1 and an atomic number of 1. We write it as:
We can symbolise individual particles in this way too; for example, a single neutron has a mass number of 1 and an atomic number of 0 (recall that the atomic number represents the charge of the nucleus, since a neutron has no charge its atomic number is 0). We write it as:
An electron has an atomic mass of 0 (since the electron is not a nucleon) and a charge of -1 (recall that the charge of an electron is equal yet opposite that of a proton). We write it as:
A proton is just the same as a hydrogen nucleus; we therefore give it the same symbol:
Isotopes
The identity of a particular atom is defined by the amount of protons it has, for example an atom with 1 proton is classified as hydrogen and an atom with 27 protons is classified as cobalt.
Although the number of protons is fixed for any classification of atom, the number of neutrons is not. This means that there can be many different variations of an atom (e.g. hydrogen), each with a different amount of neutrons; we call these variations isotopes. For example the three atoms below are all isotopes of hydrogen. They all have only 1 proton however, they each have different numbers of neutrons.
Although the number of protons is fixed for any classification of atom, the number of neutrons is not. This means that there can be many different variations of an atom (e.g. hydrogen), each with a different amount of neutrons; we call these variations isotopes. For example the three atoms below are all isotopes of hydrogen. They all have only 1 proton however, they each have different numbers of neutrons.
Types of radioactive decay
Alpha decay α |
Beta decay β |
Gamma decay 𝛾 |
|
Alpha particles α are a common byproduct of nuclear reactions. They consist of 2 protons and 2 neutrons. They are usually represented as a helium nucleus, which they are essentially identical to.
If a nucleus undergoes alpha decay... - Atomic number decreases by 2 - Atomic mass reduces by 4 |
Beta particles β are simply electrons, and are therefore represented as such. The process of beta decay involves a neutron splitting into a proton and an electron. The proton remains in the nucleus, while the electron is emitted.
If a nucleus undergoes beta decay... - Atomic number increases by 1 - Atomic mass unchanged |
Gamma radiation is an electromagnetic wave. It is essentially just energy and therefore leaves the configuration of particles in the nucleus unchanged.
If a nucleus undergoes gamma decay... - Atomic number unchanged - Atomic mass unchanged. |