Nuclear fusion

A nuclear fusion reaction involves two small "parent" nuclei combining to form a single larger "daughter" nucleus. It is essentially the opposite process of nuclear fission.
In nuclear fusion reactions, the daughter nucleus is more bound than the two parent nuclei. The daughter nucleus therefore has less energy than the two parent nuclei. This leaves an excess amount of energy, released as kinetic energy and electromagnetic radiation.
Fusion reactions require extremely high temperatures, in order to make nuclei travel fast enough to collide and fuse. For this reason, power production via terrestrial fusion is a long way away from being feasible.
In nuclear fusion reactions, the daughter nucleus is more bound than the two parent nuclei. The daughter nucleus therefore has less energy than the two parent nuclei. This leaves an excess amount of energy, released as kinetic energy and electromagnetic radiation.
Fusion reactions require extremely high temperatures, in order to make nuclei travel fast enough to collide and fuse. For this reason, power production via terrestrial fusion is a long way away from being feasible.
Riding the curve
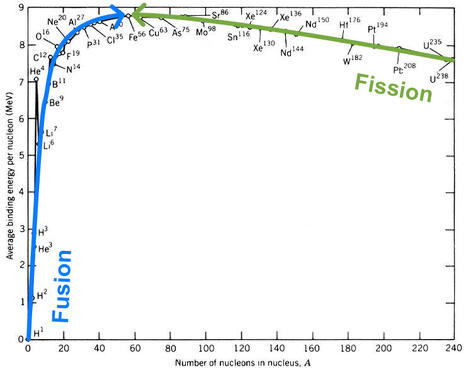
How can it be that we get energy from both fission and fusion? The answer is all about binding energy per nucleon.
For there to be a release of energy, the nuclei we end up with need to have a higher binding energy per nucleon (recall that this means less total energy) than the nuclei we started with. If you look at the graph below you will see that there are two ways to do this.
For there to be a release of energy, the nuclei we end up with need to have a higher binding energy per nucleon (recall that this means less total energy) than the nuclei we started with. If you look at the graph below you will see that there are two ways to do this.
|
The blue arrow shows the path a fusion reaction takes.
Nuclei are fused into a single larger nucleus with a higher binding energy per nucleon. Because the nucleus we end up with has a higher binding energy per nucleon than the nuclei we started with, there is an excess of energy released. |
The green arrow shows the path a fission reaction takes.
A nucleus is split into multiple smaller nuclei with a higher binding energy per nucleon. Because the nuclei we end up with have a higher binding energy per nucleon than the nucleus we started with, there is an excess of energy released. |
Stability
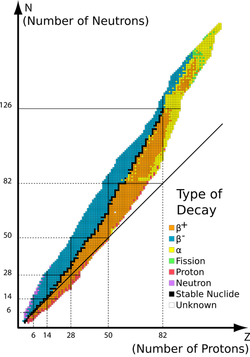
Stability refers to the proneness of a nucleus to undergo radioactive decay. The higher the binding energy per nucleon, the more stable a nucleus is.
As shown on the graph of binding energy per nucleon above, the nucleus with the highest binding energy per nucleon is Iron-56. It is the most efficiently bound nucleus.
If Iron-56 were to undergo either fission or fusion, the energy required to initiate the reaction would be greater than the energy released. In fact, fusion reactions responsible for the creation of nuclei heavier than Iron-56, must take place in supernovae (the explosions of stars).
As shown on the graph of binding energy per nucleon above, the nucleus with the highest binding energy per nucleon is Iron-56. It is the most efficiently bound nucleus.
If Iron-56 were to undergo either fission or fusion, the energy required to initiate the reaction would be greater than the energy released. In fact, fusion reactions responsible for the creation of nuclei heavier than Iron-56, must take place in supernovae (the explosions of stars).