The Bohr model
 Illustration of the Plum Pudding model
Illustration of the Plum Pudding model
The groundbreaking work of Rydberg and Balmer came at a time when our knowledge of atomic structure was still in its infancy.
At the time the popular theory of atomic structure was called the Plum Pudding model (also known as the Blueberry Muffin model), proposed by English physicist J. J. Thompson. The basic idea was that an atom was a blob of positive charge, with electrons embedded throughout.
At the time the popular theory of atomic structure was called the Plum Pudding model (also known as the Blueberry Muffin model), proposed by English physicist J. J. Thompson. The basic idea was that an atom was a blob of positive charge, with electrons embedded throughout.
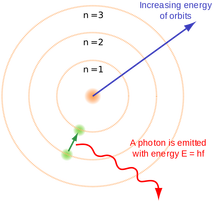 Illustration of the Bohr model
Illustration of the Bohr model
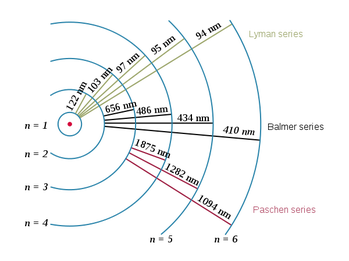
Around 15 years later a young Danish Physicist, Niels Bohr, would present a new revolutionary model of atomic structure, the so called Bohr model. This is the model taught in most classrooms today.
In the Bohr model, an atom consists of a positively charged nucleus, around which electrons orbit at specific allowed (discrete) distances.
The orbit an electron holds is related to its energy. The higher the orbit, the higher the energy an electron occupying that orbit must have.
An electron can move to a lower orbit by releasing energy, carried away as a photon of light.
Conversely, an electron can move to a higher orbit if it absorbs a photon of the right energy.
In the Bohr model, an atom consists of a positively charged nucleus, around which electrons orbit at specific allowed (discrete) distances.
The orbit an electron holds is related to its energy. The higher the orbit, the higher the energy an electron occupying that orbit must have.
An electron can move to a lower orbit by releasing energy, carried away as a photon of light.
Conversely, an electron can move to a higher orbit if it absorbs a photon of the right energy.
Energy of an electron in a specific orbit
Using the Rydberg formula, it can be shown that for a Hydrogen atom, the energy of the n-th energy level En can be given by..
Where R is Rydberg's constant, c is the speed of light, h is Planck's constant, and n is the number of the energy level, often called the quantum number.
The energy is negative because the electron is bound. In other words it has a debt of energy owed to the atom, in order for the electron to be freed from the atom, the debt of energy will need to be paid by a photon.
The first and energy level is called the ground state.
The energy is negative because the electron is bound. In other words it has a debt of energy owed to the atom, in order for the electron to be freed from the atom, the debt of energy will need to be paid by a photon.
The first and energy level is called the ground state.
Physical meaning of the Rydberg formula
Failures of the Bohr model
While the Bohr model represented a significant leap in our understanding of atomic structure, it has many failures. These are just a few of many...
In the 1920s Erwin Schrödinger would propose the Electron Cloud model, building upon the ideas proposed by a number of other great physicists of the time. It is the most widely used and accepted model of atomic structure of today.
- It does not work for atoms with multiple electrons.
- It does not explain why only certain energy levels in each atom are allowed.
- It does not explain why some energy levels are closer together than others.
In the 1920s Erwin Schrödinger would propose the Electron Cloud model, building upon the ideas proposed by a number of other great physicists of the time. It is the most widely used and accepted model of atomic structure of today.