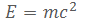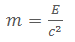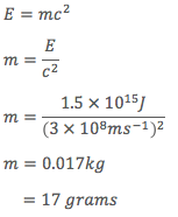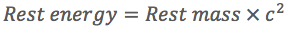Mass, matter, and energy
Mass is a quantity that describes how much an object resists acceleration. Matter exhibits mass, because of this we generally use mass as a measure of the amount of matter something has. For this reason mass and matter are sometimes incorrectly thought to be the same thing.
In 1905 Albert Einstein realised that energy also exhibits mass. The equation he used to describes how much mass energy exhibits, has become one of the most well known equations in history:
In 1905 Albert Einstein realised that energy also exhibits mass. The equation he used to describes how much mass energy exhibits, has become one of the most well known equations in history:
Where E is energy, m is the amount of mass exhibited by the energy, and c is the speed of light.
In a nuclear reaction part of the matter of a nucleus is converted into energy, reducing the amount of matter the nucleus has. This energy is then radiated away, reducing the amount of mass the nucleus has.
It is important to understand that the loss of matter and the loss of mass are separate events. Matter is lost because it is converted to energy, mass is lost because that energy (which has mass) is radiated away.
If the amount of energy radiated away is E, we would find that the mass of the nucleus will have decreased by an amount given by:
In a nuclear reaction part of the matter of a nucleus is converted into energy, reducing the amount of matter the nucleus has. This energy is then radiated away, reducing the amount of mass the nucleus has.
It is important to understand that the loss of matter and the loss of mass are separate events. Matter is lost because it is converted to energy, mass is lost because that energy (which has mass) is radiated away.
If the amount of energy radiated away is E, we would find that the mass of the nucleus will have decreased by an amount given by:
We call this the mass deficit
Example
 Photo by DB WIlson
Photo by DB WIlson
If you were to weigh the Huntly Power Station, and all the fuel and oxygen that go into it for a month, then weigh it again with all the carbon dioxide and other physical products, you would discover an unaccounted for loss of mass.
Where did this mass go? Into the New Zealand national power grid, in the form of electrical energy.
Just how much of a difference in mass would we expect? The energy output of Huntly Power Station is approximately 1.5 ⨉ 1015 Joules per month. Knowing this, we can calculate the expected mass loss per month to be...
Where did this mass go? Into the New Zealand national power grid, in the form of electrical energy.
Just how much of a difference in mass would we expect? The energy output of Huntly Power Station is approximately 1.5 ⨉ 1015 Joules per month. Knowing this, we can calculate the expected mass loss per month to be...
The energy produced by the Huntly Power Station per month, therefore has a total mass of approximately 17 grams. This also represents the amount of matter consumed to produce this energy, slightly more than is in a tablespoon of sugar.
Rest mass
Rest mass (also known as invariant mass), is defined as the mass of an object at rest. Effectively it describes the amount of matter an object has. It differs from the total mass of the object, which takes into account the mass contributed by the object's kinetic energy as well. The total mass of an object can be written as...
In a similar manner, we can define the total energy of the object as...
Where the rest energy of the object is related to its rest mass by...